This research is done in collaboration with my NSF IGERT cohort, our clients, and our affiliates.
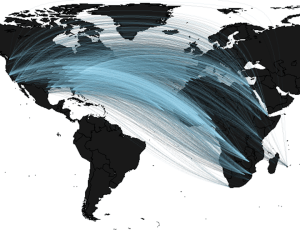
The Risk of Rift Valley Fever Introduction into the USA
We made a video and poster describing this work (as of Spring 2013) and it can be found here:
http://posterhall.org/igert2013/posters/338
The video won a national public choice award (!!) and we traveled to NSF HQ in D.C. for the award ceremony. In a nutshell- Rift Valley Fever Virus is currently endemic to Africa. The virus is transmitted between hosts by mosquitoes and has the ability to infect many different species of mammals, including humans and cattle. We are determining where the disease is most likely endemic in Africa and combining this with data on human population levels, travel behavior, and air traffic flow networks to find the relative risks of an infected human arriving at various airports across the USA.
Optimal Sampling for Early Disease Detection
The above research gave rise to some theoretical questions that we hope to answer. One of which was: Given a finite amount of resources, how do you routinely sample for a pathogen in order to detect it as early as possible after an introduction has taken place? If the pathogen is transmitted between hosts by a vector, would it be better to sample hosts, vectors, or some optimal mix of the two? Does this answer change as the disease progresses through the populations? We are answering these questions using a mix of mathematical modeling and probability theory and we hope that our results may inform groups involved in routine disease surveillance.
Published!: http://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1003668
Immunoepidemiology, Linking Within- and Between-Host Pathogen Dynamics with the Host Immune Response
What can the pathogen and immune dynamics within a host tell us about the transmission dynamics of a pathogen between hosts? When it comes to getting sick, individuals aren’t always clearly “infected” and then “recovered” as classical epidemiology models posit, but rather transition through various degrees of infection and recovery as the populations of pathogen, immune cells, and antibodies within the individuals interact. This is important when we consider how within-host dynamics affects disease spread, for instance the amount of pathogen within individuals governs the probability of the pathogen spreading to other individuals, and the rate and efficiency at which the immune system fights a pathogen impacts an individual’s recovery rate and long term immunity.
Check out some of our published work on this topic here! And here!

Pingback: COVID19 links and science | Vincent Cannataro, Ph.D.
Pingback: The Science Behind COVID-19 - Insights